Fanshawe collaborates on motion capture suit and arm sleeve for Parkinson's patients
 CREDIT: FANSHAWE COLLEGE CORPORATE COMMUNICATIONS
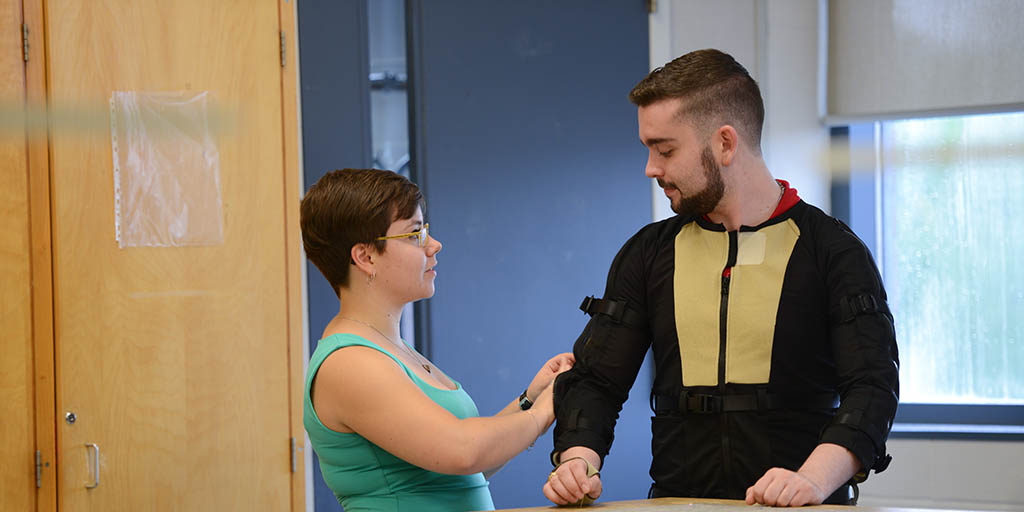
CREDIT: FANSHAWE COLLEGE CORPORATE COMMUNICATIONSFanshawe fashion design alumna, Louise Marchand, has collaborated with researchers at Movement Disorder Diagnostic Technologies Inc., in order to build a motion capture suit and tremor arm sleeve for Parkinson's patients.
Fanshawe has collaborated with researchers at Movement Disorder Diagnostic Technologies Inc. (MDDT) to create a prototype of a motion capture suit and tremor arm sleeve for Parkinson’s patients.
MDDT is a medical device company located in London, Ont., that aims to create new tools for physicians to use which will improve the quality of life for people with movement disorders.
The prototype for TremorTek, designed by recent Fanshawe fashion design graduate Louise Marchand, will contribute to more accurate reports and assessments of tremors for Parkinson’s patients.
“We wanted something easy for the patients to wear, while at the same time have our medical team guide the design of this new product. [This was] a combination of medical, engineering and design knowledge to create the perfect assessment tool,” Jack Lee, chief technology officer at MDDT, explained.
Being able to accurately assess tremors is crucial when it comes to determining the accurate dosage of Levodopa, an initial pharmacological therapy for patients with Parkinson’s.
“The TremorTek sleeve, is an experimental diagnostic device that has sensors to measure tremors and intelligently determine the treatment options available for that patient’s unique biomarker,” Lee said.
Parkinson’s is a neurogenerative disease that is caused by the death of dopamine-producing cells, which are responsible for movement by carrying signals from nerves to the brain. Symptoms of Parkinson’s include tremors, slowness and stiffness, impaired balance and rigidity of muscles.
Lee explained that despite the large variety of innovations being developed to help gain a better understanding of the disease, few people help to improve disease treatment.
“We built a Parkinson’s suit with the age, body shape and disease type of our patients in mind; a more comfortable suit for patients means a more reliable dataset for physicians to examine general Parkinson’s symptoms,” Lee said.
Marchand explained that the purpose of designing the suit was to address functionality issues with previous models.
“I initially researched many options to solve the problems presented, and through a variety of discussions with the team, as well as trial and error, we were able to come to a final design,” Marchand said.
Features of the suit include large and easy-to-use zippers and belting features, mesh underlay for breathability, anti-skid fabric to tighten the sensor pockets against the body, a two-piece design for ease of use and pockets to hold 51 sensors.
“Working with the team at MDDT was a very rewarding experience… to be able to try to improve their [Parkinson’s patients] lives in any way was a great honour,” Marchand said.
The Parkinson’s suit is currently being used as part of a clinical research project at the London Movement Disorders Centre whereas the TremorTek sleeve is waiting to be commercialized within the next year once the proper approval is received from the FDA and Health Canada.














